 Reactions with water
Reactions with water Reactions with air
Reactions with air Reactions with halogens
Reactions with halogens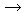 2AuCl3(s)
2AuCl3(s)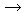 2AuBr3(s)
2AuBr3(s)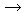 2AuI(s)
2AuI(s) Reactions with acids
Reactions with acids Reactions with bases
Reactions with bases|
Compounds | Reactions | Production
|
| Reactions of Gold |
 Reactions with water Reactions with water |
| Gold does not react with water, under any circumstances. |
 Reactions with air Reactions with air |
| Under normal conditions gold will not react with air. |
 Reactions with halogens Reactions with halogens |
| Gold reacts with chlorine and bromine to form trihalides and with iodine to form a monohalide. |
2Au(s) + 3Cl2(g)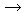 2AuCl3(s) 2AuCl3(s) |
2Au(s) + 3Br2(g)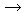 2AuBr3(s) 2AuBr3(s) |
2Au(s) + I2(g)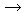 2AuI(s) 2AuI(s) |
 Reactions with acids Reactions with acids |
| Gold can be dissolved with a mixture of hydrochloric acid (HCl) and concentrated nitric acid (HNO3), in a ration of 3:1. This combination is known as aqua regia, "royal water". |
 Reactions with bases Reactions with bases |
| Gold will not react with aqueous bases. |
| Reduction Potentials | |
| Balanced half-reaction | E0 / V |
Au3+ + 2e- Au+ Au+ |
+1.41 |
Au3+ + 3e- Au(s) Au(s) |
+1.50 |
Au+ + e- Au(s) Au(s) |
+1.68 |
AuCl4- + 2e- AuCl2- + 2Cl- AuCl2- + 2Cl- |
+0.926 |
AuBr4- + 2e- AuBr2- + 2Br- AuBr2- + 2Br- |
+0.805 |
AuCl4- + 3e- Au(s) + 4Cl- Au(s) + 4Cl- |
+1.00 |
AuBr4- + 3e- Au(s) + 4Br- Au(s) + 4Br- |
+0.858 |
AuCl2- + e- Au(s) + 2Cl- Au(s) + 2Cl- |
+1.154 |
AuBr2- + e- Au(s) + 2Br- Au(s) + 2Br- |
+0.963 |